- Bentrio® remained detectable by fluorescence for up to 210 minutes in subjects' nasal passages vs. 60 minutes with saline nasal spray control
- Long nasal residence time supports extended protective effects against airborne allergens and other potentially harmful particles
HAMILTON, BERMUDA / ACCESSWIRE / March 3, 2023 / Altamira Therapeutics Ltd. (NASDAQ:CYTO), a company dedicated to developing therapeutics that address important unmet medical needs, today reported that data from a recent peer-reviewed clinical study involving Bentrio nasal spray's residence time and other rheological properties demonstrated superiority vs. a saline control.
The study results are published in Drug Development and Industrial Pharmacy (Taylor & Francis) and its full text is publicly available at: https://www.tandfonline.com/doi/full/10.1080/03639045.2023.2183724
Study excerpts:
- "The data presented in this study indicate that AM-301 (Bentrio) has a prolonged nasal residence time of up to 3.5 h compared to a saline control. AM-301 is a safe and well-tolerable medical device with features suitable for intranasal administration. Its nasal residence time and distribution encourage its use to protect nasopharyngeal areas. Two nasal spray applications per nostril provide no substantial benefit over a single spray with regards to coverage of the mucosal surface or nasal retention time."
- "Importantly, the formulation also reached the oropharynx, remaining in place for up to 240 min compared with 60 min in the control group. This feature is particularly important, since airborne viruses and allergens come into contact with both the nasal mucosa and oropharyngeal cavity."
Study Highlights
The study was performed on eight healthy volunteers that were administered Bentrio or a classic saline nasal spray marked with a fluorescent agent (fluoresceine 0.1%) to track the distribution within the nasal cavity and the nasal residence time for up to 240 minutes. Participants were administered a single dose or, in case of Bentrio, also a repeated dose at a different spray angle. Bentrio was found to be widely distributed in the lower to middle parts of the nasal cavity (in particular inferior turbinate and septum, to a lesser extent also on the middle turbinate), which is where airborne allergen particles typically collect. The thin protective film was present for 210 minutes. There were no meaningful differences between single and repeated dose application of Bentrio, confirming that one puff is sufficient.
In contrast, saline nasal spray showed a shorter nasal residence time of 60 minutes only. Bentrio's extended presence within the nasal cavity is achieved thanks to its special thixotropic properties: the formulation is a gel designed to adhere to the nasal mucosa for an extended time, whereas a saline solution is prone to rapid run-off. Bentrio's gel formulation becomes sprayable upon shaking it briefly prior to application. Bentrio's long nasal residence time is supporting the extended protective effect of the nasal spray as it could be observed in other studies, including grass pollen and house dust mite challenge studies.
Images
In the single dose saline spray control group, fluorescence disappeared within 60 minutes from the nose, whereas the groups using a single dose of Bentrio retained a detectable amount of fluorescence for up to 210 minutes on the middle and inferior turbinates. As shown in Figure 1- Anterior Rhinoscopy below, images were taken at different time points to track the amount of fluorescence still visible after a single application of Bentrio or saline nasal spray.
Figure 1
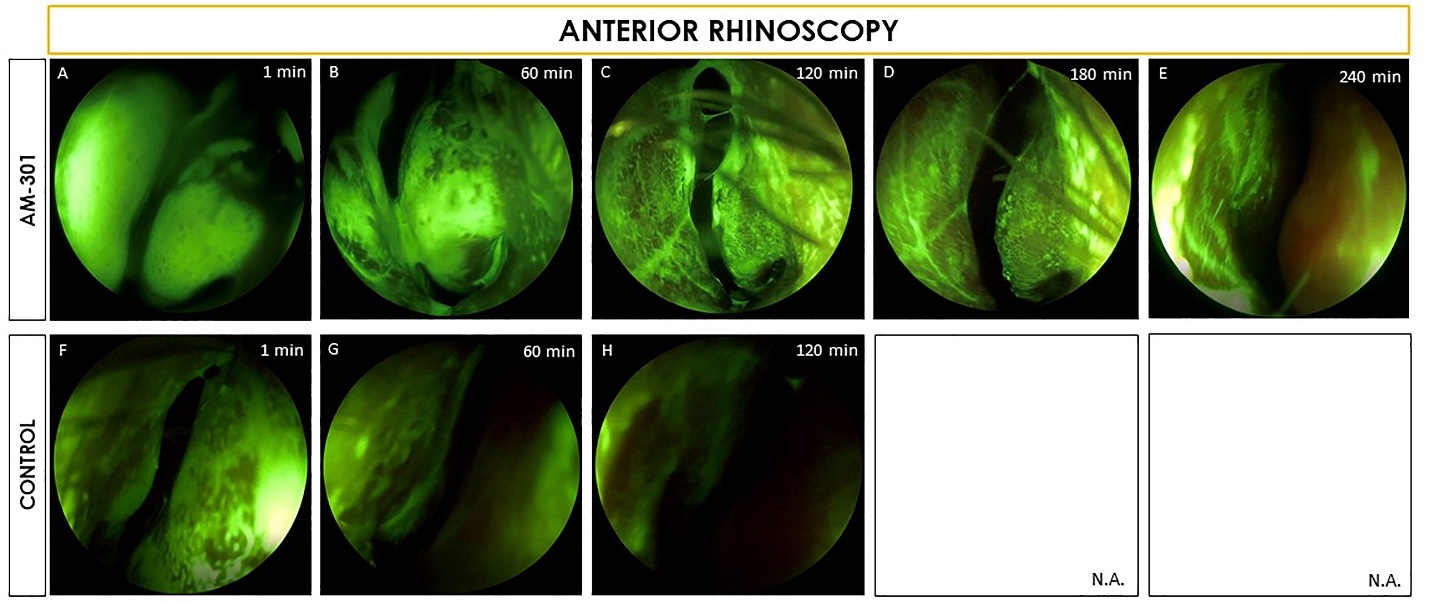
While nasal residence time was the main focus of the study, the presence of the fluorescence in the oropharynx was monitored for 240 minutes as well. The image below, Figure 2- Oropharyngeal Endoscopy, shows that fluorescence for all three groups started to appear in the throat five to ten minutes after administration. The finding that Bentrio does not slow down discharge from the nasal cavity to the throat, suggests that the protective film established by Bentrio within the nasal cavity does not interfere with mucociliary clearance, a normal, physiological process that helps the body to clear particles out of the nose for discharge via the digestive tract. The saline control group's fluorescence disappeared after 90-120 minutes versus the Bentrio groups which showed fluorescence for up to 240 minutes.
Figure 2

The results of the study show that the rheological properties of the AM-301 formulation allow it to be sprayed once and effectively provide extended coverage and protection of the nasal mucosa without interfering with natural nasal clearance.
The study was conducted and examinations performed at the Department of Otorhinolaryngology, Head and Neck Surgery of the University Medical Center of the Eberhard-Karls University Tübingen, Germany.
About Drug Development and Industrial Pharmacy
The aim of Drug Development and Industrial Pharmacy is to publish novel, original, peer-reviewed research and review manuscripts related to pharmaceutical science research, specifically in the fields of nanomedicines, formulation, development and delivery technologies, biopharmaceuticals, new material development and processing technology advancement. Research papers must be hypothesis driven and emphasize innovative breakthrough topics. Timely critical review papers within the journal scope are also welcomed.
About Altamira Therapeutics
Altamira Therapeutics (Nasdaq:CYTO) is dedicated to developing RNA-based therapeutics for extrahepatic targets (OligoPhore™ / SemaPhore™ delivery platforms). The Company currently has two flagship siRNA programs in preclinical development beyond in vivo proof of concept: AM-401 for KRAS driven cancer and AM-411 for rheumatoid arthritis. The versatile delivery platform is also suited for mRNA and other types of RNA therapeutics and shall be leveraged via out-licensing to pharma or biotech companies. In addition, Altamira is in the process of divesting and/or licensing-out its legacy assets in allergology and viral infection (Bentrio® OTC nasal spray; commercial) and inner ear therapeutics (AM-125 nasal spray for vertigo; post Phase 2; Keyzilen® and Sonsuvi® for tinnitus and hearing loss; Phase 3). Founded in 2003, Altamira is headquartered in Hamilton, Bermuda, with its main operations in Basel, Switzerland. For more information, visit: https://altamiratherapeutics.com/
Forward-Looking Statements
This press release may contain statements that constitute "forward-looking statements" within the meaning of Section 27A of the Securities Act of 1933 and Section 21E of the Securities Exchange Act of 1934. Forward-looking statements are statements other than historical facts and may include statements that address future operating, financial or business performance or Altamira Therapeutics' strategies or expectations. In some cases, you can identify these statements by forward-looking words such as "may", "might", "will", "should", "expects", "plans", "anticipates", "believes", "estimates", "predicts", "projects", "potential", "outlook" or "continue", or the negative of these terms or other comparable terminology. Forward-looking statements are based on management's current expectations and beliefs and involve significant risks and uncertainties that could cause actual results, developments and business decisions to differ materially from those contemplated by these statements. These risks and uncertainties include, but are not limited to, the success of the continued commercialization of Bentrio and success of strategic transactions, including licensing or partnering, with respect to Bentrio or any other legacy assets, Altamira Therapeutics' need for and ability to raise substantial additional funding to continue the development of its product candidates, the timing and conduct of clinical trials of Altamira Therapeutics' product candidates, the clinical utility of Altamira Therapeutics' product candidates, the timing or likelihood of regulatory filings and approvals, Altamira Therapeutics' intellectual property position and Altamira Therapeutics' financial position, including the impact of any future acquisitions, dispositions, partnerships, license transactions or changes to Altamira Therapeutics' capital structure, including future securities offerings. These risks and uncertainties also include, but are not limited to, those described under the caption "Risk Factors" in Altamira Therapeutics' Annual Report on Form 20-F for the year ended December 31, 2021, and in Altamira Therapeutics' other filings with the SEC, which are available free of charge on the Securities Exchange Commission's website at: www.sec.gov. Should one or more of these risks or uncertainties materialize, or should underlying assumptions prove incorrect, actual results may vary materially from those indicated. All forward-looking statements and all subsequent written and oral forward-looking statements attributable to Altamira Therapeutics or to persons acting on behalf of Altamira Therapeutics are expressly qualified in their entirety by reference to these risks and uncertainties. You should not place undue reliance on forward-looking statements. Forward-looking statements speak only as of the date they are made, and Altamira Therapeutics does not undertake any obligation to update them in light of new information, future developments or otherwise, except as may be required under applicable law.
CONTACT
Hear@altamiratherapeutics.com
800-460-0183
SOURCE: Altamira Therapeutics Ltd.
View source version on accesswire.com:
https://www.accesswire.com/741841/Clinical-Study-Demonstrates-Bentrios-Superior-Nasal-Residence-Time-and-Rheological-Properties