Spruce Biosciences Announces Integrated Long-Term Clinical Data of Tralesinidase Alfa Enzyme Replacement Therapy (TA-ERT) Demonstrating Profound and Durable Efficacy and Safety in Patients with Sanfilippo Syndrome Type B (MPS IIIB)
TA-ERT Profoundly and Durably Lowered Cerebral Spinal Fluid Heparan Sulfate Non-Reducing End (CSF HS-NRE), the Factor Responsible for Neurodegeneration in MPS IIIB
U.S. FDA Confirmed that CSF HS-NRE is a Surrogate Biomarker Reasonably Likely to Predict Clinical Benefit and Could Serve as Basis for Accelerated Approval
Cognition and Cortical Grey Matter Volume Were Stabilized in Patients Treated with TA-ERT Relative to Declines Observed in Untreated Patients
Spruce Biosciences, Inc. (OTCQB: SPRB), a late-stage biopharmaceutical company focused on developing and commercializing novel therapies for neurological disorders with significant unmet medical need, today announced results from a long-term data integration of tralesinidase alfa enzyme replacement therapy (TA-ERT) clinical program in patients with Sanfilippo Syndrome Type B (MPS IIIB).
This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20250814678929/en/
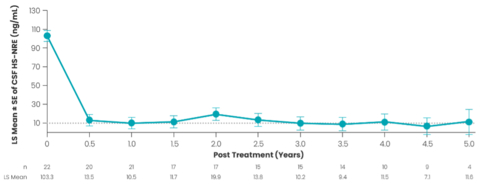
Figure 1. TA-ERT Therapy Leads to Profound and Durable Reduction in CSF HS-NRE Over a 5-Year Period. Dashed line represents upper limit of normal (10 ng/mL) for CSF HS-NRE; LS = least squares; LS mean ±SE based on mixed-model repeated measures (MMRM) analysis with study visit as covariate. Note: CSF HS-NRE levels were averaged over each 0.5-year interval.
Spruce integrated and evaluated group-level efficacy data for cerebral spinal fluid heparan-sulfate non-reducing end (CSF HS-NRE), cortical grey matter volume (CGMV), and Bayley-III Cognitive Raw Score (BSID-C), the cognitive subscale of the Bayley Scales of Infant and Toddler Development - Third Edition (Bayley-III), as well as safety data over a five-year period from clinical studies 201, 202, and 401. Data from treated patients (n=22) in the studies 201, 202, and 401 was compared with data from untreated patients with MPS IIIB in natural history studies 901 and 902.
“These data demonstrate a rapid, profound, and durable effect of ICV-administered TA-ERT on normalizing CSF HS-NRE, the pathogenic factor leading to neurodegeneration, and in stabilizing CGMV and cognitive function, relative to the declines observed in untreated children with Sanfilippo Syndrome Type B (MPS IIIB) in the natural history studies,” said Paul Harmatz, M.D., Principal Investigator in studies 901, 201, and 202 and Professor in Residence, Department of Pediatrics, University of California, San Francisco (UCSF) and UCSF Benioff Children’s Hospital Oakland. “This is an important development toward relief for children and families whose lives are impacted by this devastating condition.”
“Convincing positive outcomes have been experienced by families who resoundingly state that the benefits of TA-ERT outweigh risks in their real-world experiences,” said Cara O’Neill, M.D., FAAP, Co-Founder and Chief Science Officer of Cure Sanfilippo Foundation. “Biomarker data, along with promising clinical outcomes, are significant and meaningful from the perspective of the patient community. Currently, there is no U.S. FDA-approved therapy for the treatment of MPS IIIB, and disease management consists of limited palliative care. We are eager to see TA-ERT advance under the accelerated approval pathway.”
Cerebral Spinal Fluid Heparan-Sulfate Non-Reducing End (CSF HS-NRE)
Integrated group-level data from clinical studies 201, 202, and 401 demonstrate that TA-ERT therapy significantly reduced to normal or near normal CSF HS-NRE levels over a five-year period (Figure 1). At 240 weeks, CSF HS-NRE decreased 91.5 ng/mL from baseline (95% CI: -102.10, -80.90; p<0.0001). Most participants experienced normalization of CSF HS-NRE levels eight weeks after initiating therapy. In a 2024 meeting with the U.S. Food and Drug Administration (FDA), the FDA confirmed that CSF HS-NRE is a surrogate biomarker reasonably likely to predict clinical benefit and could serve as a basis for accelerated approval.
Cognitive Function
In untreated patients with MPS IIIB, cognitive function peaks at around four years of age and then declines over time. In contrast, children with established disease treated with TA-ERT experienced stable cognitive function over time (Figure 2). Untreated children in the natural history studies showed a decline in cognition beginning at approximately five years of age that progressively worsened over time, while cognition in the TA-ERT treated group remained stable. Using a model-based approach, the mean (95% CI) BSID-C over six to 10 years of age was significantly higher in patients treated with TA-ERT, relative to untreated, age-matched children, with differences evident at six years of age (group difference: 10.67, 95% CI: 3.23, 18.11; p=0.005). At 10 years of age, the difference in BSID-C scores between groups increased to 34.66 (95% CI: 24.38, 44.93; p<0.0001). Although TA-ERT treatment stabilized BSID-C scores on average, increases in BSID-C scores were more commonly observed in subjects who initiated therapy at younger ages with higher baseline cognitive function and prior to the establishment of meaningful neurodegeneration. The BSID-C is anticipated to be the primary endpoint for the post-marketing clinical trial.
Cortical Grey Matter Volume (CGMV)
TA-ERT therapy was also associated with stabilization of CGMV, relative to the decline in CGMV observed in untreated children due to the progressive neurodegenerative nature of MPS IIIB (Figure 3). While CGMV should increase with age in children up to five years of age, there was an average loss of ~32 mL over 48 weeks in untreated children with MPS IIIB observed in study 901. Consistent with TA-ERT’s mechanism of action, decreases in CGMV were observed during the initial 24 weeks of TA-ERT treatment, likely reflecting intracellular clearance of cerebral spinal fluid heparan sulfate (CSF HS) and CSF HS-NRE. CGMV stabilized from weeks 48 to 240 with TA-ERT treatment.
Safety
TA-ERT therapy exposure for up to 7.3 years has demonstrated an adequate safety profile in a serious and fatal disease for which no treatment is currently available. The mean (SD) exposure to TA-ERT was 4.2 (2.0) years. No deaths occurred throughout study 201 and its long-term extension studies 202 and 401. The most frequent treatment-emergent adverse event (TEAE) by preferred term (reported in ≥40% of participants) was vomiting (22 [100%]), followed by pyrexia (20 [90.9%]), upper respiratory tract infection (17 [77.3%]), pleocytosis (11 [50.0%]), COVID-19 infection (10 [45.5%]), and diarrhea (9 [40.9%]). Four (18%) patients discontinued treatment, although three (14%) discontinuations were due to hydrocephalus, a known complication of MPS IIIB. Adverse events related to the ICV device were consistent with other therapies administered by the ICV route.
About Sanfilippo Syndrome Type B (MPS IIIB)
MPS IIIB is an ultra-rare, serious, and fatal genetic disease characterized by deficiency in N-Acetyl-Alpha-Glycosaminidase (NAGLU), an enzyme required for the catabolism of heparan sulfate (HS) in lysosomes. It is estimated that MPS IIIB affects fewer than 1:200,000 people in the United States (U.S.), but the true incidence and prevalence are difficult to ascertain because MPS IIIB is a disease currently not included in newborn screening. The accumulation of toxic levels of cerebral spinal fluid heparan sulfate in the brain is the underlying pathophysiology of MPS IIIB. Although signs and symptoms of MPS IIIB can vary amongst affected individuals, progressive neurodegeneration typically follows a predictable path to brain atrophy, cognitive and developmental impairment, hyperactivity with aggressive and destructive behavior, delayed speech, hearing loss, and motor skill deficits. Somatic manifestations include coarse facial features, hepatosplenomegaly, and gastrointestinal symptoms. The final stage of MPS IIIB is typically marked by severe dementia, loss of motor function, and seizure activity, with patients largely bed-ridden and requiring constant care, requiring feeding tubes for hydration and nutrition, and ultimately leading to death. The estimated life expectancy of individuals with MPS IIIB ranges from 15 to 19 years of age. Currently, there are no FDA-approved therapies for MPS IIIB, and management of the disease consists of limited palliative care to improve quality of life.
About Tralesinidase Alfa Enzyme Replacement Therapy (TA-ERT)
Tralesinidase Alfa Enzyme Replacement Therapy (TA-ERT) is a fusion protein comprised of recombinant human alpha-N-acetylglucosaminidase (rhNAGLU). TA-ERT is intended as an enzyme replacement therapy for the treatment of patients with MPS IIIB (Sanfilippo Syndrome Type B) who lack rhNAGLU enzyme activity. TA-ERT is expected to restore rhNAGLU enzyme activity in the central nervous system following intracerebroventricular injection. rhNAGLU typically lacks the mannose-6-phosphate residues that are essential for efficient cellular uptake via the M6P receptor pathway. As a result, the naked enzyme is poorly absorbed by cells, including neurons. To address this challenge, TA-ERT is fused to an insulin-like growth factor 2 peptide, which binds to the cation-independent mannose-6-phosphate on cell surfaces. This fusion enables the enzyme to be internalized and delivered to the lysosome, thereby enhancing its therapeutic potential for treating MPS IIIB. By restoring NAGLU enzymatic activity and promoting clearance of lysosomal heparan sulfate and heparan sulfate non-reducing end in the brain, TA-ERT therapy is expected to preserve neuronal cell health and potentially halt or slow the neurological decline and improve clinical outcomes in affected patients. TA-ERT has been evaluated in three clinical studies in participants with MPS IIIB: the interventional study 201 and extension studies 202 and 401. Twenty-two individuals with MPS IIIB have been administered TA-ERT therapy. TA-ERT has demonstrated an adequate safety profile based on integrated five years of safety data.
About Spruce Biosciences
Spruce Biosciences is a late-stage biopharmaceutical company focused on developing and commercializing novel therapies for neurological disorders with significant unmet medical need. To learn more, visit www.sprucebio.com and follow us on X, LinkedIn, Facebook and YouTube.
Forward-Looking Statements
Statements contained in this press release regarding matters that are not historical facts are “forward-looking statements” within the meaning of the Private Securities Litigation Reform Act of 1995. Such forward-looking statements include statements regarding, among other things, the ability to seek accelerated approval of TA-ERT for MPS IIIB based on existing clinical data. Because such statements are subject to risks and uncertainties, actual results may differ materially from those expressed or implied by such forward-looking statements. Words such as “anticipate”, “will”, “potential”, “intend”, “expect” and similar expressions are intended to identify forward-looking statements. These forward-looking statements are based upon Spruce’s current expectations and involve assumptions that may never materialize or may prove to be incorrect. Actual results could differ materially from those anticipated in such forward-looking statements as a result of various risks and uncertainties, which include, without limitation, risks and uncertainties associated with Spruce’s business in general, the impact of geopolitical and macroeconomic events, and the other risks described in Spruce’s filings with the U.S. Securities and Exchange Commission. All forward-looking statements contained in this press release speak only as of the date on which they were made and are based on management’s assumptions and estimates as of such date. Spruce undertakes no obligation to update such statements to reflect events that occur or circumstances that exist after the date on which they were made, except as required by law.
View source version on businesswire.com: https://www.businesswire.com/news/home/20250814678929/en/
Contacts
Media
Katie Beach Oltsik
Inizio Evoke Comms
(937) 232-4889
Katie.Beach@inizioevoke.com
media@sprucebio.com
Investors
Samir Gharib
President and CFO
Spruce Biosciences, Inc.
investors@sprucebio.com
More News
View More



Recent Quotes
View More
Quotes delayed at least 20 minutes.
By accessing this page, you agree to the Privacy Policy and Terms Of Service.