The International Registry is an observational, multicentre, non-interventional program for patients diagnosed with non-melanoma skin cancer (NMSC), to inform treatment outcomes and patterns of care at international hospitals.
MUNICH - Feb. 14, 2022 - PRLog -- OncoBeta® GmbH is pleased to announce that the first patients have registered into the world-first International Registry that is capturing data-driven treatment results for NMSC treatments.
The first-of-its kind International Registry was launched by OncoBeta via specialist health solutions partner Avion Medical in November 2021 to collect real-world patient data for three mainstay treatments of NMSC. Its goal is to identify optimal methods of treating the disease that can be applied internationally.
The International Registry is an easy-to-use platform for physicians and will capture data observing how treatments and innovative approaches influence long-term patient outcomes. The Registry also has an important focus on patient-reported outcomes such as quality of life and comfort of treatment.
The first patients have been recorded into the online registry that follows the treatment journey, from the initial disease assessment to treatments and ongoing assessments. Not only that, but there is an opportunity for patients to contribute their feedback and record their experiences via a questionnaire that includes impact on quality of life as well as comfort score through the OncoBeta WeBe mobile app.
Dr Gerhard Dahlhoff, Medical Director at OncoBeta, says, "To date, no unified reportage on NMSC and its treatments is available to the international medical community. The Registry aims to rectify this shortfall in our understanding of the disease treatment. The WeBe app has been designed so it is easy to use for the patient and the physician, which is critical in ensuring participation and the delivery of robust data. The patient data collection will provide key insights from the patient's perspective about their treatment and the disease."
An international committee made up of multi-disciplinary clinicians from South Africa, Australia, Italy and Germany is leading the data collection and outputs of the registry. Paolo Castellucci, Nuclear Medicine Physician at S. Orsola, Bologna, and chairman of the International Registry, says, "I'm excited to be involved in an International Registry that will be the first to capture robust, long-term data that will reveal patterns of care, identify which treatment provides the best long term outcomes for patients."
Shannon D Brown III, CEO at OncoBeta GmbH, says, "We are thrilled to see the first patients being registered in the International Registry. By working with clinicians around the world, the Registry will draw from a larger sample size to identify important insights and establish best practices in the treatment of NMSC."
Participation in the International Registry for Non-Melanoma Skin Cancer Treatment is by invitation only. Physicians that would like to become part of the Registry and offer Rhenium-SCT treatment at your centre, contact info@oncobeta.com
About the Rhenium-SCT® (Skin Cancer Therapy)
Non-melanoma skin cancer (NMSC) is the most common form of cancer in humans. The most common cause of NMSC is sun exposure, while other predisposing factors include genetic skin conditions and immunosuppressive diseases or treatments.1
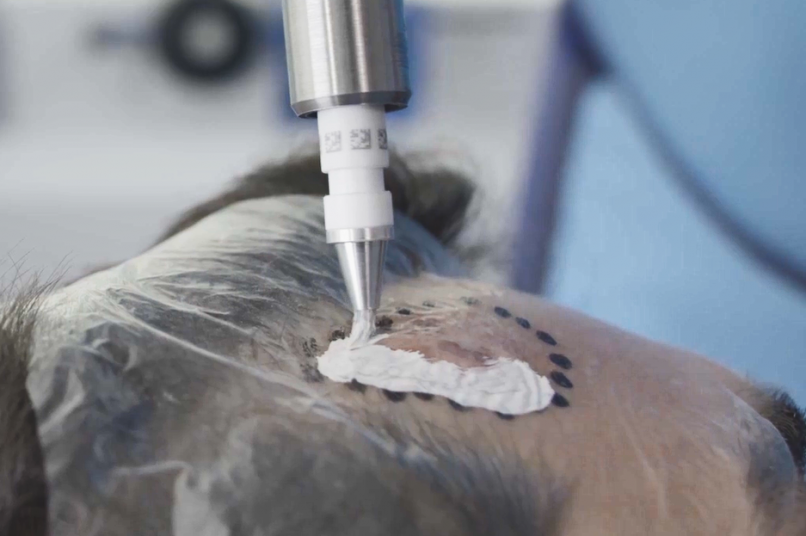
The Rhenium-SCT® is a painless*, single session†, non-invasive therapy providing for unparalleled aesthetic results, even in cases otherwise considered difficult to treat.2-4 The Rhenium-SCT utilizes the radioisotope Rhenium-188 in an epidermal application with optimal properties for the treatment of NMSCs (non-melanoma skin cancers). The Rhenium-SCT is a precise, personalised therapy that is only applied to the area needed to treat without affecting the healthy tissue. The specially designed device ensures the Rhenium-SCT compound never comes in direct contact with the patient's skin and the application is safe and simple for the applying physician. Most cases of NMSCs (Basal Cell Carcinomas and Squamous Cell Carcinomas) can be treated using the Rhenium-SCT in one single session†4. Scar-free healing4 of the treated lesion area and the regeneration of healthy tissue occurs usually within a few weeks after treatment4.
About OncoBeta® GmbH
OncoBeta® GmbH, with its headquarters located in Garching near Munich, Germany, is a privately held medical device company, specializing in the development and commercialization of state-of-the-art, innovative therapies. Since its foundation, OncoBeta has concentrated its efforts on the development, regulatory approval(s) and commercialization of the epidermal radioisotope therapy Rhenium-SCT® (Skin Cancer Therapy), targeting NMSCs. OncoBeta has perfected the customized application and device management system in conformity with all health, safety and environmental protection regulatory standards.
Find out more about the Rhenium-SCT® at www.oncobeta.com
Follow us on social media:
LinkedIn: https://www.linkedin.com/company/oncobeta-gmbh/
Facebook: https://www.facebook.com/oncobeta/
Instagram: https://www.instagram.com/oncobeta_gmbh/
About Avion Medical
Avion Medical is a specialist health solutions partner, providing data management and clinical strategy consulting, specialising in Trials and Registries, for pharma, biotech and medical devices. Avion Medical's proprietary platform AviData Clinical Cloud enables seamless integration between patient inputs, imaging, lab data, eCRF and reporting; whilst ASTON Safety Reporting provides a comprehensive, web-based pharmacovigilance platform and database for processing and storing drug, device and vaccine adverse events. Avion Medical delivers specialised solutions for clinical research and product commercialisation.
Find out more about Avion Medical at www.avionmedical.com.au
Forward-looking statements
This announcement includes forward-looking statements that involve risks, uncertainties and other factors, many of which are outside of OncoBeta's control, and which could cause actual results to differ materially from the results discussed in the forward-looking statements. Forward-looking statements include statements concerning OncoBeta's plans, objectives, goals, future events, performance and/or other information that is not historical information. All such forward-looking statements are expressly qualified by these cautionary statements and any other cautionary statements which may accompany the forward-looking statements. OncoBeta undertakes no obligation to publicly update or revise forward-looking statements to reflect subsequent events or circumstances after the date made, except as required by law.
*No reported pain2,3
†Complete tumour regression in 98.5% of lesions treated, with 89% after a single application4
References
- Cancer.net. Skin Cancer (Non-Melanoma): Risk Factors and Prevention. October 2020. https://www.cancer.net/cancer-types/skin-cancer-non-melanoma/risk-factors-and-prevention (accessed October 2021).
- Cipriani C, et al. J Dermatolog Treat. 2020; Jul 22:1-7.
- Sedda AF, et al. Clin Exp Dermatol. 2008;33(6):745-749.
- Cipriani C, Sedda AF. Epidermal Radionuclide Therapy - Dermatological High-Dose-Rate. Brachytherapy for the Treatment of Basal and Squamous Cell Carcinoma. In: Therapeutic Nuclear Medicine, editor Baum RP; New York: Springer, 2014.
Contact
Jane Morey
***@moreymedia.com.au
Photos: (Click photo to enlarge)

Read Full Story - First Patients Registered Into OncoBeta's International Registry For Non-Melanoma Skin Cancer Treatments | More news from this source
Press release distribution by PRLog