Respiratory Pathogen Testing kits Market is evolving into a value-driven ecosystem, where clinical utility, workflow integration, and data connectivity define competitive advantage.
NEWARK, DE / ACCESS Newswire / May 4, 2026 / According to the latest market analysis by Future Market Insights The global respiratory pathogen testing kits market is moving beyond pandemic-era volatility into a more structured, protocol-led growth phase-where institutional procurement, clinical accuracy, and operational efficiency matter more than sheer testing volume. Hospitals, laboratories, and public health systems are standardizing respiratory diagnostics workflows, embedding multiplex molecular testing into routine care pathways. This shift is quietly reshaping vendor selection, pricing models, and long-term demand visibility across the diagnostics industry.
Quick Stats at a Glance
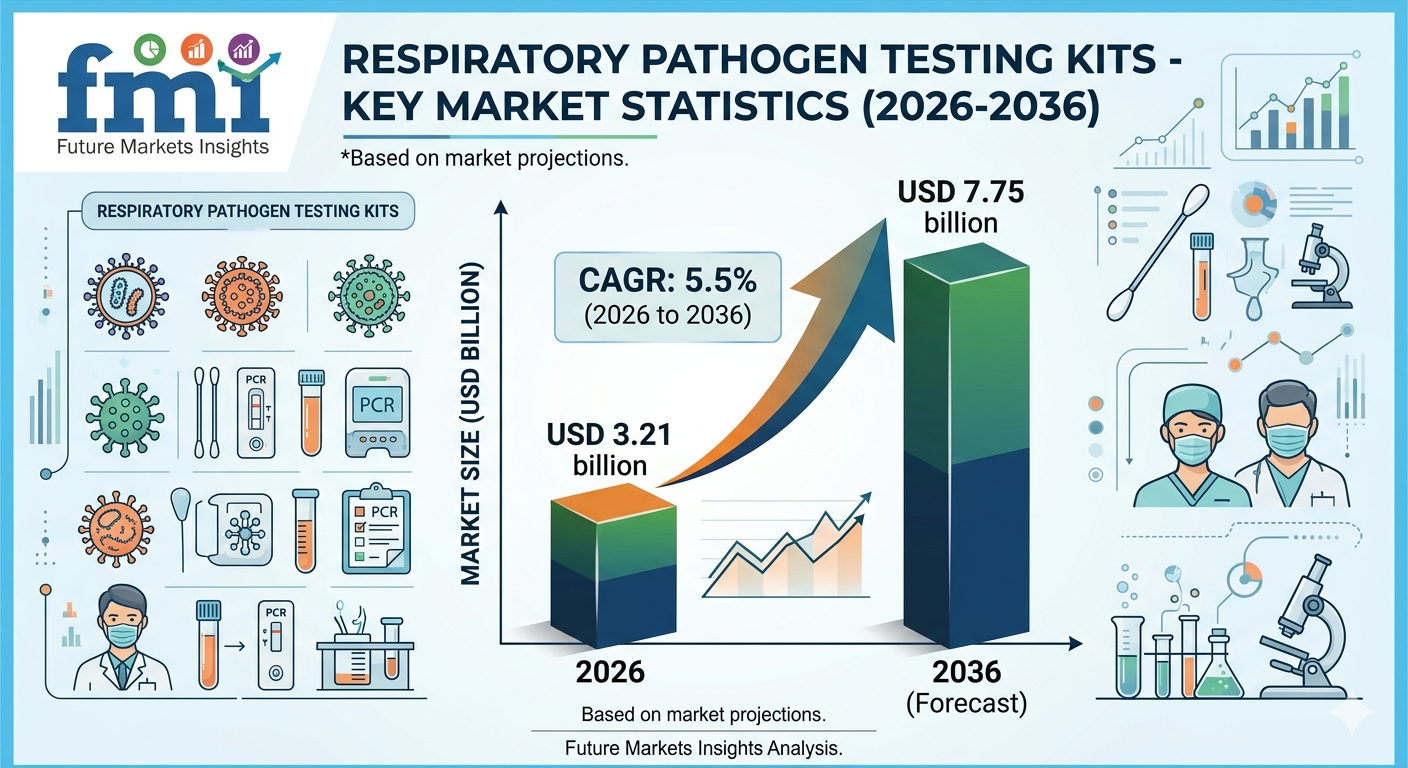
2026 Market Value: (2026): USD 3.21 billion
2036 Forecast Value: USD 7.75 billion
CAGR (2026-2036): 5.5%
Leading Product Segment (2026): RT-PCR kits (45.0% share)
Leading Technology: NAAT (58.0% share)
Top Growth Markets: USA (5.7%), European Union (5.6%)
Get detailed market forecasts, competitive benchmarking, and pricing trends: https://www.futuremarketinsights.com/reports/sample/rep-gb-10542
Market Size and Forecast: From Surge Demand to Sustained Utilization
Between 2026 and 2036, the respiratory pathogen testing kits market is projected to grow steadily at a CAGR of 5.5%, reaching USD 7.75 billion. Unlike the pandemic years, growth is now underpinned by recurring, protocol-based procurement cycles rather than emergency-driven purchasing.
Healthcare systems are integrating respiratory testing into seasonal preparedness strategies-particularly for influenza, RSV, and emerging coronavirus variants-creating predictable, recurring revenue streams for manufacturers.
Growth Drivers: Surveillance, Speed, and Syndromic Testing
Three structural drivers are sustaining market expansion:
Institutional Surveillance Mandates Government-backed programs led by organizations like the Centers for Disease Control and Prevention and the European Centre for Disease Prevention and Control are enforcing continuous monitoring of respiratory diseases. This ensures baseline demand for high-throughput diagnostic platforms.
Rise of Syndromic (Multiplex) Panels Hospitals increasingly favor single-test panels capable of detecting multiple pathogens-such as influenza A/B, RSV, and SARS-CoV-2-from one sample. This reduces diagnosis time and improves clinical decision-making.
Expansion of Point-of-Care Testing Retail pharmacies, outpatient clinics, and home testing channels are accelerating adoption of rapid antigen and decentralized diagnostic kits, broadening the addressable market beyond traditional labs.
Challenges: Cost Pressures and Regulatory Complexity
Despite stable growth, the market faces tangible constraints:
High Cost per Test: Molecular diagnostics remain expensive, limiting penetration in price-sensitive markets across Asia, Africa, and Latin America.
Reimbursement Gaps: In many emerging economies, lack of insurance coverage restricts adoption outside hospital settings.
Tightening Regulations: Frameworks like EU IVDR and evolving FDA clearance pathways are increasing compliance costs and time-to-market.
These factors are reinforcing a two-tier market structure-advanced molecular diagnostics in developed regions and cost-driven immunoassays in emerging markets.
Opportunities: Decentralization and AI Integration
The next wave of opportunity lies in accessibility and intelligence:
Home-Based Testing: Growing consumer acceptance is opening new revenue streams for rapid kits.
AI-Enabled Diagnostics: Companies like Roche Diagnostics are integrating AI-assisted interpretation tools to improve turnaround time and accuracy.
Cloud-Connected Surveillance: Real-time data sharing is enhancing outbreak tracking and public health responsiveness.
Segmentation Insights: Molecular Dominance with Expanding Use Cases
By Product: RT-PCR kits dominate with a 45% share in 2026, driven by their high sensitivity and ability to support multiplex testing.
By Technology: NAAT leads with 58% share, reflecting institutional preference for molecular-level detection accuracy.
By End User: Hospitals account for 49% of demand, as acute care settings require rapid and reliable diagnostics for treatment decisions and infection control.
By Infection Type: Key applications include influenza, human coronavirus, and pneumonia diagnostics-areas where rapid identification significantly impacts clinical outcomes.
Customize insights for your business strategy@ https://www.futuremarketinsights.com/customization-available/rep-gb-10542
Regional Analysis: Developed Markets Drive Standardization
United States (5.7% CAGR) The U.S. leads global growth due to strong reimbursement frameworks, CDC-backed surveillance, and widespread adoption of multiplex panels.
European Union (5.6% CAGR) Demand is shaped by cross-border disease monitoring and regulatory harmonization under IVDR, favoring large, compliant manufacturers.
Asia (Japan & South Korea at 5.5%) High diagnostic testing rates and robust public health infrastructure support steady growth, though cost sensitivity persists in broader Asia-Pacific markets.
Emerging Markets
Adoption remains uneven due to infrastructure gaps and pricing constraints, with immunoassay-based kits dominating.
Competitive Landscape: Scale, Compliance, and Multiplex Capability Win
The market is moderately consolidated, with global leaders competing across technology platforms and geographic reach:
Abbott Laboratories leads with ~18% share, leveraging its strength in rapid antigen and integrated diagnostics.
Thermo Fisher Scientific and Roche Diagnostics dominate high-throughput molecular testing.
bioMerieux differentiates through syndromic panel innovation.
QuidelOrtho Corporation focuses on decentralized and rapid testing solutions.
Barriers to entry remain high due to regulatory requirements, capital-intensive manufacturing, and entrenched hospital procurement relationships.
Strategic Implications for Industry Stakeholders
For Manufacturers: Investment in multiplex panels, cost optimization, and regulatory compliance is critical to securing long-term contracts.
For Healthcare Providers: Transition toward standardized testing protocols requires balancing accuracy, turnaround time, and cost per result.
For Investors: The shift to recurring demand models improves revenue predictability, making diagnostics a more stable healthcare segment.
For Procurement Leaders: Vendor selection is increasingly based on total cost of ownership and panel breadth, not just unit price.
Unlock 360° insights for strategic decision making and investment planning: https://www.futuremarketinsights.com/checkout/10542
Future Outlook: From Volume to Value
The respiratory pathogen testing kits market is evolving into a value-driven ecosystem, where clinical utility, workflow integration, and data connectivity define competitive advantage. Growth will not be explosive-but it will be durable, predictable, and strategically important.
Multiplex diagnostics, decentralized testing, and AI integration will define the next decade, while regulatory rigor will continue to favor established players.
Executive Takeaways
The market is shifting from pandemic-driven demand to structured, protocol-based procurement.
Multiplex molecular testing is becoming the standard in hospital settings.
Cost and reimbursement challenges will shape adoption in emerging markets.
Decentralized testing and AI integration represent the next major growth frontier.
Companies that balance accuracy, affordability, and regulatory readiness will capture the most value through 2036
Read More Related Report Form Future Market Insight (FMI)
Babesiosis Treatment Market: https://www.futuremarketinsights.com/reports/babesiosis-treatment-market
Eisenmenger Complex Management Market: https://www.futuremarketinsights.com/reports/eisenmenger-complex-management-market
Central Pain Syndrome Management Market: https://www.futuremarketinsights.com/reports/central-pain-syndrome-management-market
PD1 Non-Small Cell Lung Cancer Treatment Market: https://www.futuremarketinsights.com/reports/pd1-non-small-cell-lung-cancer-treatment-market
Klinefelter Syndrome Therapeutics Market: https://www.futuremarketinsights.com/reports/klinefelter-syndrome-therapeutics-market
About Future Market Insights (FMI)
Future Market Insights, Inc. (FMI) is an ESOMAR-certified, ISO 9001:2015 market research and consulting organization, trusted by Fortune 500 clients and global enterprises. With operations in the U.S., UK, India, and Dubai, FMI provides data-backed insights and strategic intelligence across 30+ industries and 1200 markets worldwide.
Contact Us:
Rahul Singh
AVP - Marketing and Growth Strategy
Future Market Insights, Inc.
+91 8600020075
For Sales - sales@futuremarketinsights.com
For Media - Rahul.Singh@futuremarketinsights.com
For web - https://www.futuremarketinsights.com/
SOURCE: Future Market Insights, Inc.
View the original press release on ACCESS Newswire




