LONDON - Sept. 27, 2022 - PRLog -- OncoBeta® GmbH today announced King's College Hospital in London, United Kingdom, is the latest medical facility to participate in the global phase IV EPIC-Skin Study (Efficacy of Personalised Irradiation with Rhenium-SCT – for the treatment of non-melanoma skin cancer [NMSC]).
The first United Kingdom patients were treated with Rhenium-SCT® at King's College are among 210 adults participating in the international study that will follow their progress over the next 24 months. The EPIC-Skin study is also being conducted through study centres located in Australia, Austria and Germany.
Dr Nicola Mulholland, Consultant Nuclear Medicine Physician at King's College Hospital and study chief investigator says, "With rates of NMSC on the rise around the world, it's vitally important that new treatments are developed and tested. We are very pleased to be able to offer new therapies to patients involved in studies and trials, with the hope of advancing treatment for skin cancer."
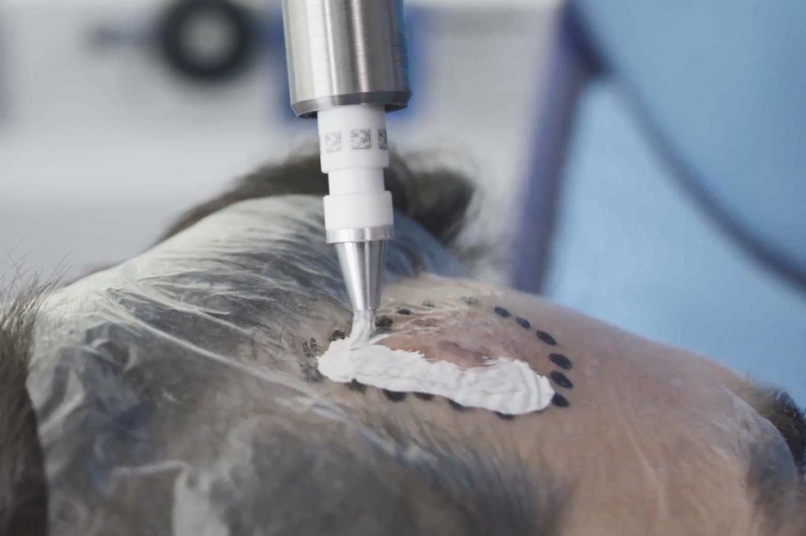
Global incidence rates of NMSCs are increasing, with more than 7.7 million cases of NMSC being recorded each year.1,2 Standard treatments for NMSCs are surgery-based, which can risk scarring or loss of function. Rhenium-SCT® uses a non-invasive paste containing ß-emitting particles directly to the lesion, which target cancer cells without the need for surgery, in one session.3-5
Dr Saqib Bashir, Consultant Dermatologist and Skin Cancer Lead at King's College Hospital says, "When it comes to cancer research, it's important to continually innovate. This study offers an opportunity to further investigate the efficacy of non-invasive epidermal radioisotope therapy."
The EPIC-Skin study will measure patient reported outcomes such as quality of life, treatment comfort and cosmetic outcomes, as well as further evaluating the efficacy of Rhenium-SCT® for the treatment of NMSC. Patients in the study will be able to record their experiences via a user-friendly clinical study app.
Dr Gerhard Dahlhoff, Medical Director at OncoBeta GmbH, says, "Rhenium-SCT® allows for a targeted and non-invasive treatment of NMSCs without damaging adjacent healthy tissue. It is important we collect data through the EPIC-Skin Study to further validate the effectiveness of Rhenium-SCT®, and the role it plays as a patient-friendly treatment alternative."
Clinicians who are interested in enrolling patients in the study can contact OncoBeta directly at www.oncobeta.com/contact.
About the Rhenium-SCT® (Skin Cancer Therapy)
Non-melanoma skin cancer (NMSC) is the most common form of cancer in humans.2 The most common cause of NMSC is sun exposure, while other predisposing factors include genetic skin conditions and immunosuppressive diseases or treatments.6
The Rhenium-SCT® is a painless*, single session†, non-invasive therapy that provides aesthetic results, even in cases otherwise considered difficult to treat.3-5 The Rhenium-SCT utilizes the radioisotope Rhenium-188 in an epidermal application with optimal properties for the treatment of NMSCs (non-melanoma skin cancers). The Rhenium-SCT is a precise, personalised therapy that is only applied to the area needed to treat without affecting the healthy tissue. The specially designed device ensures the Rhenium-SCT compound never comes in direct contact with the patient's skin and the application is safe and simple for the applying physician. Most cases of NMSCs (Basal Cell Carcinomas and Squamous Cell Carcinomas) can be treated using the Rhenium-SCT in one single session.†5 Scar-free healing of the treated lesion area and the regeneration of healthy tissue occurs usually within a few weeks after treatment.5
About OncoBeta®
OncoBeta®, with its headquarters located in Garching near Munich, Germany, is a privately held medical device company, specializing in the development and commercialization of state-of-the-art, innovative therapies. Since its foundation, OncoBeta has concentrated its efforts on the development, regulatory approval(s) and commercialization of the epidermal radioisotope therapy Rhenium-SCT® (Skin Cancer Therapy), targeting NMSCs. OncoBeta has perfected the customized application and device management system in conformity with all health, safety and environmental protection regulatory standards.
Find out more about the Rhenium-SCT® at www.oncobeta.com
Follow us on social media:
LinkedIn: www.linkedin.com/company/oncobeta-gmbh/
Facebook: www.facebook.com/oncobeta/
Instagram: www.instagram.com/oncobeta_gmbh/
Forward-looking statements
This announcement includes forward-looking statements that involve risks, uncertainties and other factors, many of which are outside of OncoBeta's control, and which could cause actual results to differ materially from the results discussed in the forward-looking statements. Forward-looking statements include statements concerning OncoBeta's plans, objectives, goals, future events, performance and/or other information that is not historical information. All such forward-looking statements are expressly qualified by these cautionary statements and any other cautionary statements which may accompany the forward-looking statements. OncoBeta® undertakes no obligation to publicly update or revise forward-looking statements to reflect subsequent events or circumstances after the date made, except as required by law.
*No reported pain3,4
†Complete tumour regression in 98.5% of lesions treated.5
References
- Global Burden of Disease Cancer Collaboration, et al. JAMA Oncol. 2019;5(12):1749-1768.
- Ciążyńska M, et al. Sci Rep. 2021;11(1):4337.
- Cipriani C, et al. J Dermatolog Treat. 2020; Jul 22:1-7.
- Sedda AF, et al. Clin Exp Dermatol. 2008;33(6):745-749.
- Cipriani C, Sedda AF. Epidermal Radionuclide Therapy - Dermatological High-Dose-Rate. Brachytherapy for the Treatment of Basal and Squamous Cell Carcinoma. In: Therapeutic Nuclear Medicine, editor Baum RP; New York: Springer, 2014.
- Cancer.net. Skin Cancer (Non-Melanoma): Risk Factors and Prevention. October 2020. https://www.cancer.net/cancer-types/skin-cancer-non-melanoma/risk-factors-and-prevention (accessed March 2022).
Contact
Jane Morey
***@moreymedia.com.au
Photos: (Click photo to enlarge)
Read Full Story - London skin cancer patients undergo first treatments with OncoBeta's Rhenium-SCT as part of the EPIC-Skin study | More news from this source
Press release distribution by PRLog

